De nombreuses études ont étudié le lien potentiel entre les champs électromagnétiques d’extrêmement basses fréquences (CEM-EBF) et la santé au cours des 40 dernières années. L’exposition « court terme/intensité élevée » est bien étudiée chez les volontaires humains et les animaux. Les effets observés ont des seuils en deçà desquels ils ne se produisent pas et peuvent être évités en respectant les restrictions de base appropriées (champs électriques induits dans le corps).
Des questions se posent quant à l’exposition « long terme/faible intensité ». Les CM-EBF sont classés par le CIRC comme “potentiellement cancérigènes pour l’homme” sur base de données épidémiologiques montrant un risque accru de leucémie infantile. D’autres études soulèvent la question du rôle des CEM 50 Hz dans le développement du cancer, de la maladie d’Alzheimer et d’autres maladies. Cependant, les études ne permettent pas de conclure et des recherches approfondies sont nécessaires.
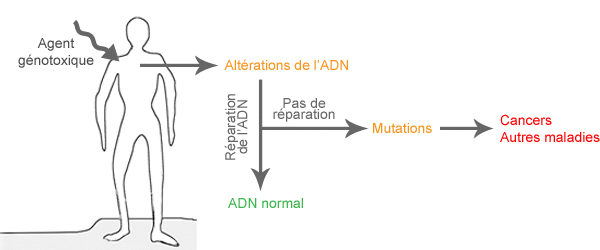
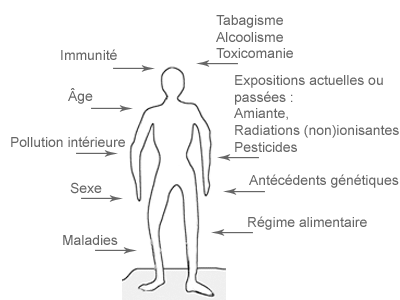
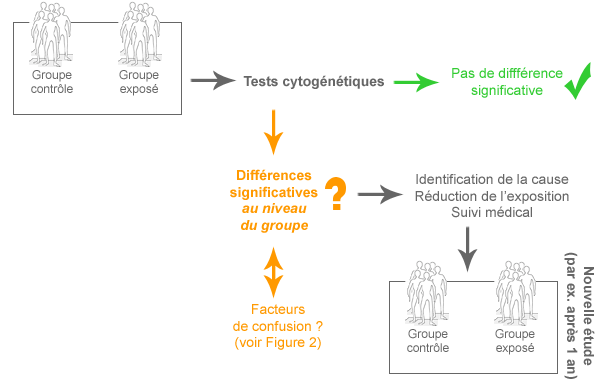
Il est donc important de déterminer les conséquences biologiques dans des groupes particulièrement exposés, comme par exemples les travailleurs exposés aux CEM. Une étude de biomonitoring cytogénétique est utile pour déterminer si les CEM-EBF peuvent induire des dommages génétiques.
Qu’est-ce qu’une étude de biomonitoring cytogénétique et comment pourrait-elle aider à comprendre les effets des CEM sur la santé ? Voici comment une telle étude est menée et comment elle doit être interprétée.