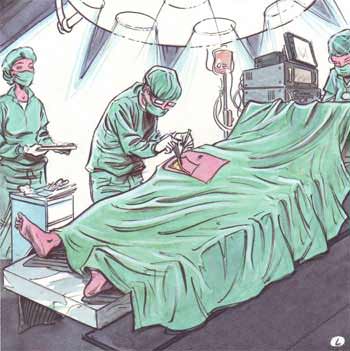
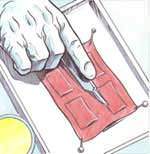
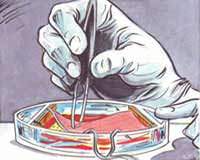
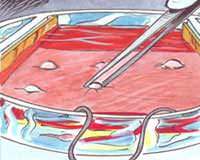
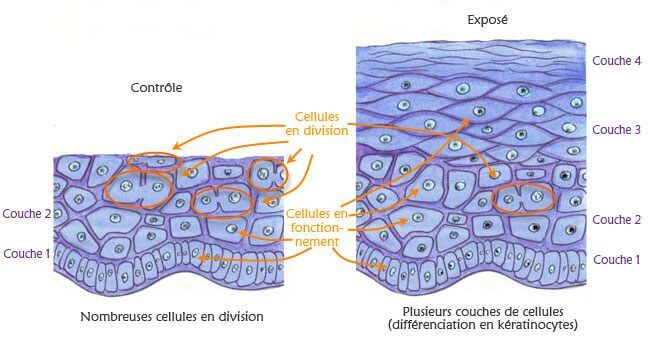
This study intends to analyse the biological effects of extremely low frequency electric and magnetic fields. Keratinocytes, cells of the outermost layer of the human skin (the epidermis), are placed on a piece of decellularized dermis, the inner layer of the human skin. Two platinum electrodes are used to apply the electric signal. Gene activation is compared in exposed cells and in sham exposed cells.
This model of epidermal repair and electric signal provides simplified and well characterized model to study the biological effects of electromagnetic stimulation.
Here is a presentation of the study and the main results.